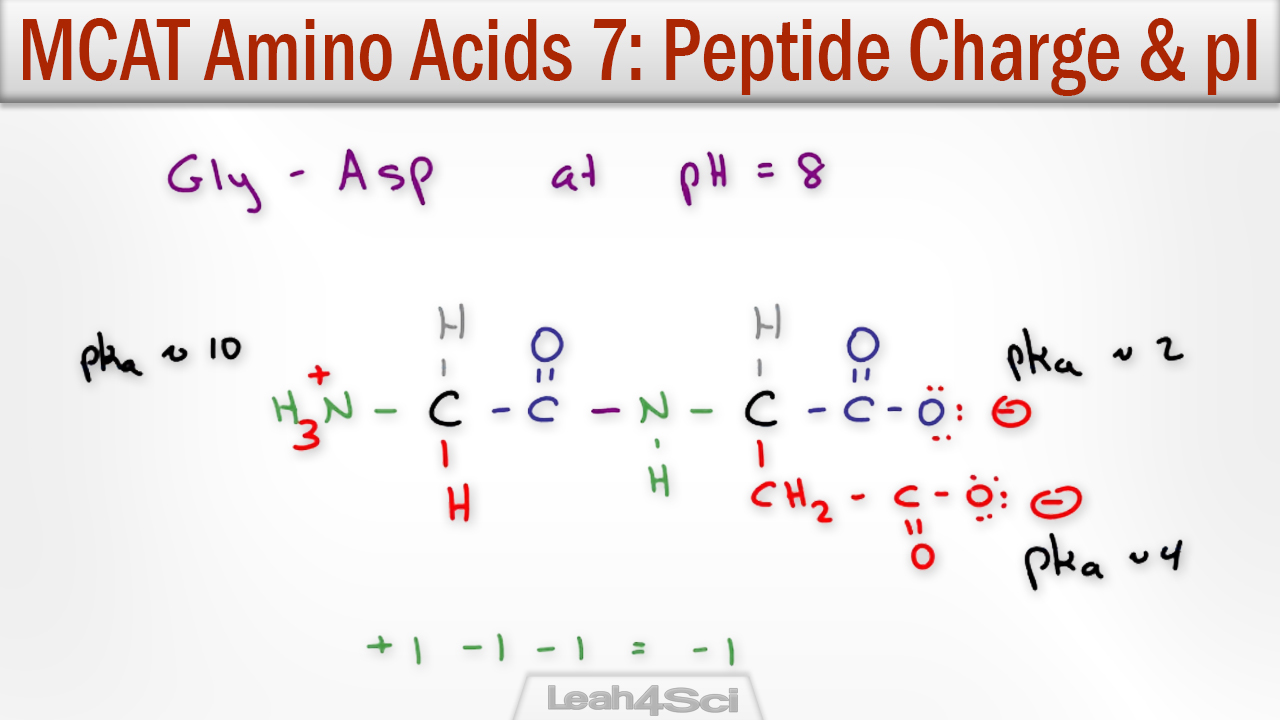
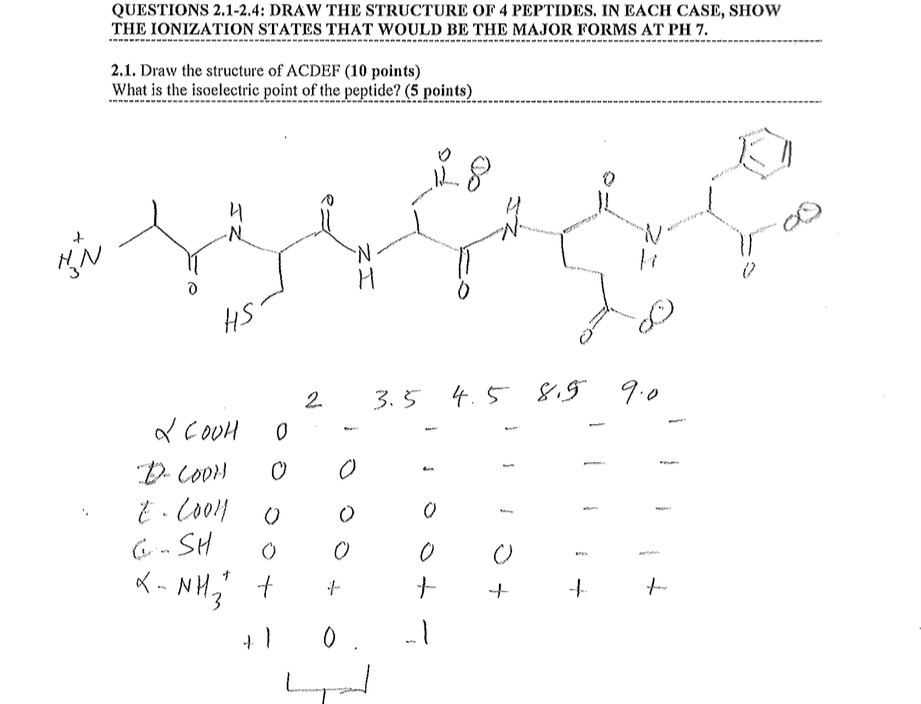
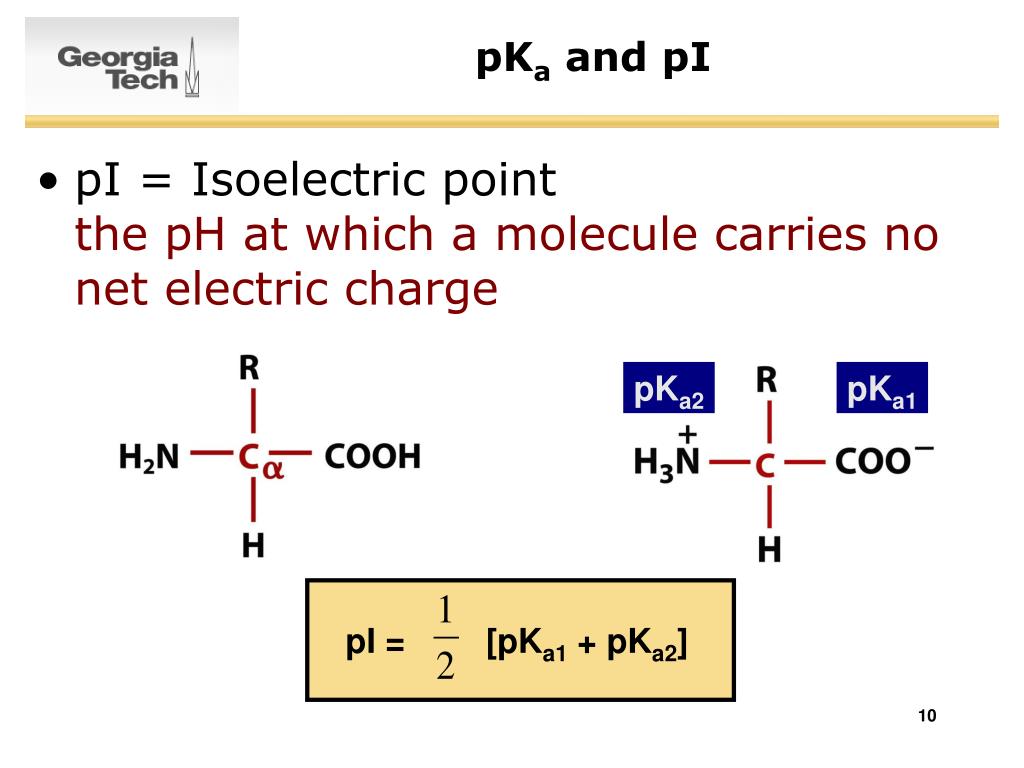
Unlike the water molecule, these net charges are independent of each other. On the other side, we have a carboxyl group which let go of a proton therefore gaining a -1 net charge (negative). In the case of the amino acids shown in the video above, we have an amino group which gained a proton from the solution and its net charge became + 1 (positive). At this point, the molecule is polar, additionally there are no formal charges. The oxygen atom "Hogs" the electrons from the covalent bond creating a partially negative side in the molecule while the other side becomes partially positive. Therefore, summing the charges at pH 7, the net charge comes to 0.I thought of that too, but my guess would be no.Ī water molecule is considered polar because of electronegativity differences between the oxygen atom and the 2 hydrogen atoms. The following table estimates the pH: pKa Let's see what would the net charge be at pH 7 The next step is the estimation of a pH where the net charge would be 0 upon summation. Ths side chain of Cysteine (Cys) would have a pKa value of 8.37 Ths side chain of Lysine (Lys) would have a pKa value of 10.54 Ths side chain of the 2 Glutamic Acid (Glu) would have a pKa value of 4.07 The carboxylic terminal of the peptide would carry a pKa of 1.9 The amino terminal of the peptide would carry a pKa of 9.6 The pKa on the ionizable group is then noted: To calculate the isoelectric point (pI) of a peptide, the individual amino acids are first considered. Molecules are capable of ionization, therefore, they can gain or lose electrons thereby resulting in a positive or a negative charge on the chemical species respectively. The isoelectric point (pI) can be defined as the point where molecules such as an amino acid carry no net electrical charge at a given pH (potential of hydrogen).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
